To guard against this, by-pass diodes need to be integrated into the solar cell. All GaAs-based multi-junction cells are also vulnerable to so-called 'reverse voltage bias' – when part of the array is covered by shadow, as might occur during a manoeuvre or a particular orbital season then electric surges can damage the array. If a bare cell was exposed to the space environment it would degrade as much within a few days as a protected cell does in 15 years. To minimise the harmful effects of space radiation – mainly energetic electrons and protons – all solar cells are covered by cover glass, typically just 100 micrometres (0.1 mm) thick. Through such design strategies, current solar cells on geostationary satellites still retain 88% of their original performance after 15 years resulting in an absolute end-of-life efficiency of 24.6%." "The trick is to find the right combination of materials and doping levels to ensure all junctions are perfectly matched at the end of the satellite's life. And each of these layers needs to be closely aligned to achieve an optimal cell."ĭesigning the connections between cell junctions therefore requires a great deal of expertise and innovation, he adds: "All layers must be matched regarding the current flowing through the cells to avoid efficiency losses, even though the junctions have different current density capabilities, and which change over time because space radiation degrades the individual junctions at a different rate. "They are all needed to monolithically integrate the three power-generating junctions and to ensure that the maximum amount of light enters the solar cell and is not reflected at the surface. "In practice each triple-junction solar cell has about 30 individual film layers," says Mr Gerlach. The resulting leap in efficiency from single to triple junction cells is on the order of ten percentage points, although designing and building them is hard. Forming a single overall structure, these layers are stacked in series based on their sensitivity: the topmost junction reacts to highest-energy blue light while letting other light through the middle layer absorbs medium-energy green light then the bottom layer makes its contribution from the remaining lowest-energy red light. These allow the placing of multiple junctions on top of one another, each tuned to different segments of the overall light spectrum. 
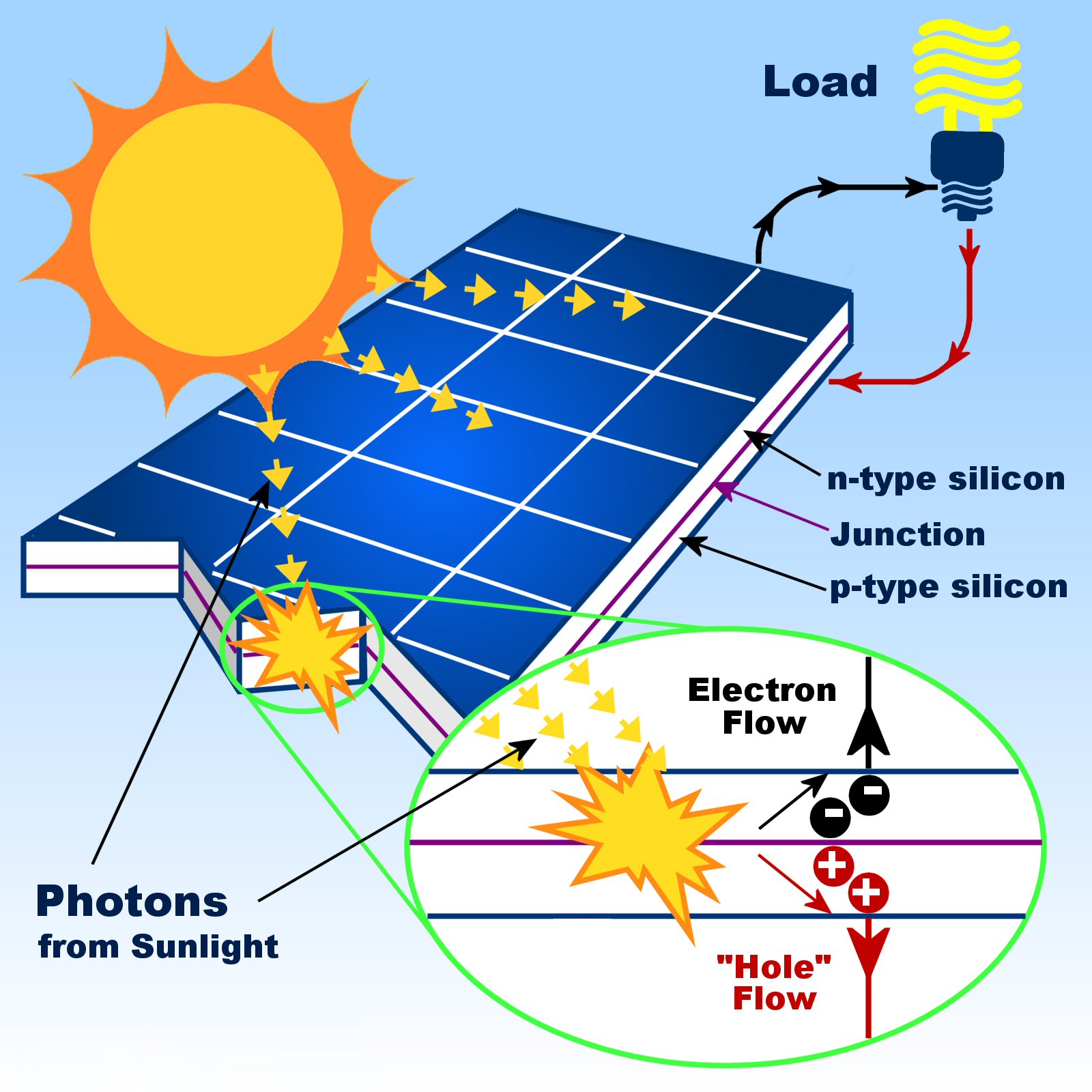
If you connect the n-type and p-type layers with a metallic wire, the electrons will travel from the n-type layer to the p-type layer by crossing the depletion zone and then go through the external wire back of the n-type layer, creating a flow of electricity.Silicon was once the semiconductor of choice but it is increasingly supplanted by gallium arsenide (GaAs) –based semiconductors which reach higher power levels. If this happens in the electric field, the field will move electrons to the n-type layer and holes to the p-type layer. When sunlight strikes a solar cell, electrons in the silicon are ejected, which results in the formation of “holes”-the vacancies left behind by the escaping electrons. The presence of these oppositely charged ions creates an internal electric field that prevents electrons in the n-type layer to fill holes in the p-type layer. When all the holes are filled with electrons in the depletion zone, the p-type side of the depletion zone (where holes were initially present) now contains negatively charged ions, and the n-type side of the depletion zone (where electrons were present) now contains positively charged ions. This creates an area around the junction, called the depletion zone, in which the electrons fill the holes (Fig. Near the junction of the two layers, the electrons on one side of the junction (n-type layer) move into the holes on the other side of the junction (p-type layer). In the n-type layer, there is an excess of electrons, and in the p-type layer, there is an excess of positively charged holes (which are vacancies due to the lack of valence electrons). Instead, it is free to move inside the silicon structure.Ī solar cell consists of a layer of p-type silicon placed next to a layer of n-type silicon (Fig. It bonds with its silicon neighbor atoms, but one electron is not involved in bonding. Phosphorus has five electrons in its outer energy level, not four. 
The n-type silicon is made by including atoms that have one more electron in their outer level than does silicon, such as phosphorus. Because boron has one less electron than is required to form the bonds with the surrounding silicon atoms, an electron vacancy or “hole” is created. The p-type silicon is produced by adding atoms-such as boron or gallium-that have one less electron in their outer energy level than does silicon. A solar cell is made of two types of semiconductors, called p-type and n-type silicon. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
